
Das securPharm-System
Aufbau als End-to-End System
Das securPharm-System basiert auf dem Ende-zu-Ende-Prinzip, bei dem die beiden Enden der Logistikkette zur Absicherung dienen. Dabei ist ein Ende der pharmazeutische Unternehmer, der ein Arzneimittel in Verkehr bringt. Das andere Ende ist die Abgabestelle, etwa eine öffentliche Apotheke.
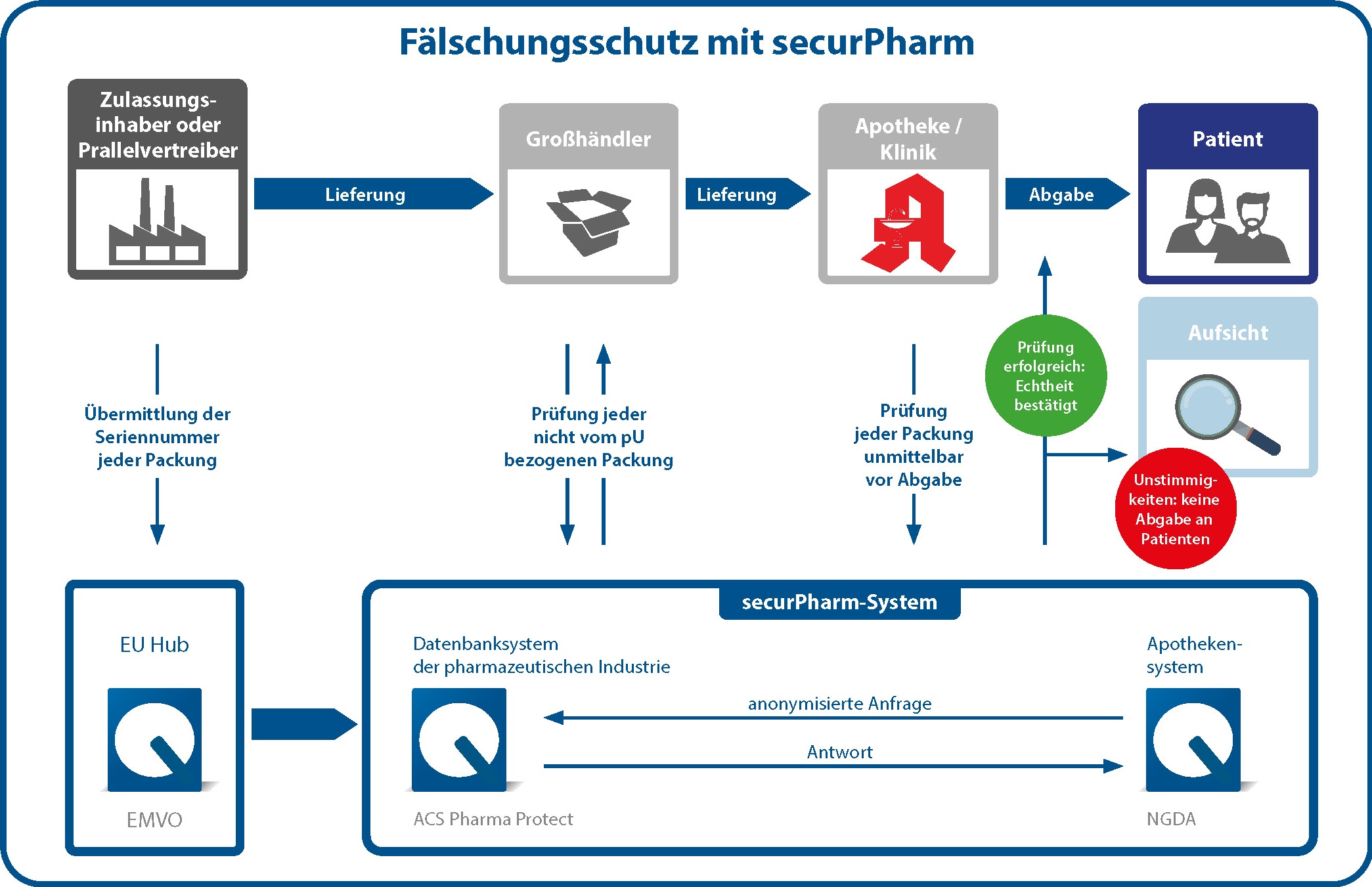
Das deutsche Verifikationssystem ist eingebettet in ein europäisches Netzwerk, um auch grenzüberschreitend den Patientenschutz zu gewährleisten. Der EU-Hub, betrieben von der European Medicines Verification Organisation (EMVO), sorgt für den Datenaustausch zwischen den Mitgliedstaaten der EU, des EWR und der Schweiz.
Ablauf der Echtheitsprüfung
Während des Produktionsprozesses versieht der pharmazeutische Unternehmer jede Packung eines Arzneimittels mit den Sicherheitsmerkmalen. Die Daten des individuellen Erkennungsmerkmals (Seriennummer, Produktcode, Chargennummer, Verfalldatum) werden in Klarschrift und im Data Matrix Code auf die Packung aufgebracht und in die zentrale Datenbank der pharmazeutischen Industrie hochgeladen. Zur Echtheitsprüfung wird der Data Matrix Code vor der Abgabe an den Patienten gescannt. Dadurch werden die Daten der Packung mit den Daten im System abgeglichen. Der Status der Packung wird an die Apotheke zurückgemeldet, d.h. ob das individuelle Erkennungsmerkmal aktiviert oder bereits deaktiviert wurde. Ist letzteres der Fall – etwa weil die Packung mit diesem Merkmal bereits abgeben wurde – darf die Packung nicht an den Patienten abgegeben werden. Der Vorfall wird dann überprüft. Ist das Erkennungsmerkmal aktiviert, darf das Arzneimittel abgegeben werden.
Wie schützt das System gegen Fälschungen?
Das europäische Schutzsystem macht Arzneimittelfälschungen in der legalen Lieferkette leichter erkennbar, da jede Seriennummer nur einmal genutzt werden kann. Über die Seriennummer in Kombination mit dem Produktkode und der Chargennummer ist außerdem rück-verfolgbar, wo ein gefälschtes Arzneimittel in die legale Lieferkette eingetreten ist. Im Falle von gestohlener Ware kann die Seriennummer im System gesperrt werden, so dass die Ware nicht mehr abgegeben werden kann.
Strikte Datentrennung
Aus Gründen des Datenschutzes arbeitet das securPharm-System mit getrennten Systemen für pharmazeutischen Unternehmen (Datenbanksystem der pharmazeutischen Industrie) und die Arzneimittel-abgebenden Stellen (Apothekensystem). Verifikationsanfragen werden über das Apothekensystem gebündelt und anonymisiert an das Datenbanksystem der pharmazeutischen Industrie weitergeleitet. Dabei werden die Teilsysteme von unseren strategischen Partner NGDA (Apothekensystem) und ACS (Datenbanksystem der pharmazeutischen Industrie) betrieben.
Ansprechpartner finden
Haben Sie Fragen oder brauchen Sie Hilfe? Unsere Übersicht hilft Ihnen, den richtigen Ansprechpartner zu finden. Dieser kann Ihnen dann weiterhelfen. Behörden wenden sich bitte direkt an den securPharm e.V.
Kontakt aufnehmen